Rechargeable Batteries
In recent years lithium-ion batteries (LIBs) have become the dominant technology for powering a range of electrical devices, finding applications in areas ranging from portable consumer electronics to electric vehicles and even grid storage. As economies around the world rush to decarbonise, faced with issues such as climate change, worsening air pollution and the finite nature of fossil fuels, rechargeable batteries have emerged as a key enabling technology to aid the green transition. However, while dramatic progress has been made in improving the performance of lithium-ion batteries over the last 30 years, they still suffer from issues such as high cost, safety concerns, limited energy densities, charging times and lifetimes. These challenges necessitate further development of novel electrode materials (both anodes and cathodes) which can overcome these drawbacks and further increase the utilisation of LIBs in high performance applications.
As a complement to LIBs, sodium-ion batteries (NIBs) have gained increasing research attention over the last 10 years due to attractive factors such as low cost, enhanced safety profile and reliance on non-toxic and abundant elements. However, due to the larger ionic radius and higher redox potential of Na, NIBs typically suffer from significantly lower energy densities compared to LIBs. This makes them attractive in stationary applications such as grid storage and power back-up systems, where high safety and low cost take priority over weight and volume. Never-the-less, full commercialisation of NIBs requires significant improvements in performance to maximise the energy density, power capability and lifetime, whilst maintaining high safety and low cost.
To achieve this, in our group, we investigate a range of electrode materials for applications in LIBs and NIBs to move these technologies beyond the current state-of-the-art and enable the next generation of batteries to match the demands of future applications. Our studies focus on materials exhibiting anion redox; low-cost sodium-based cathode materials; oxalate-based cathodes (including those exhibiting anion redox), and organic anode materials.
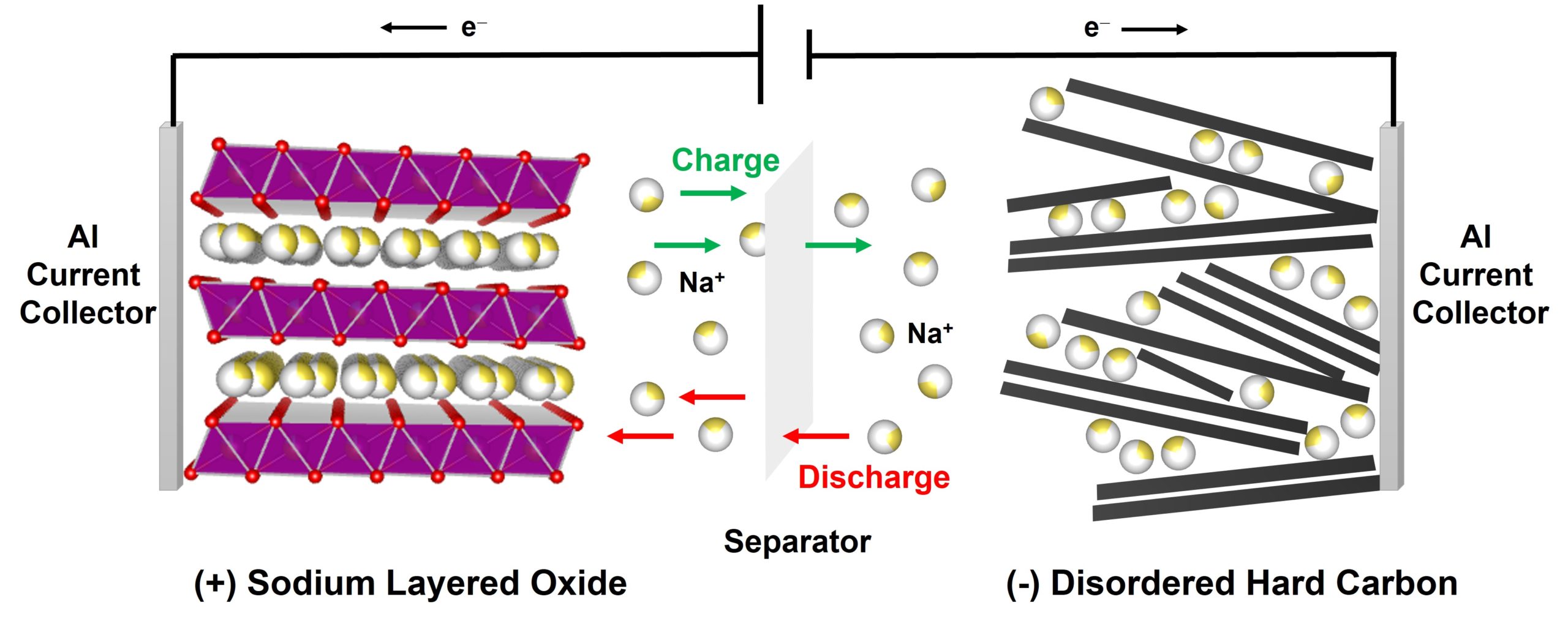
To evaluate the structural and electronic structural changes on charge/discharge in these battery systems, we use a variety of laboratory-based and synchrotron/neutron diffraction and spectroscopic methods in combination with advanced microscopy and electrochemical techniques. These fundamental studies allow us to understand the complex structure-property-performance relationships which govern overall battery performance and enable the development of novel high-performance materials.